C. Lionne, M. Gelin, G. Labesse
The goal of the group is to help the design of efficient and specific drugs by studying enzyme reaction pathways and drug-target interactions by the mean of fine enzymology, biophysical characterization and thermodynamic approaches.
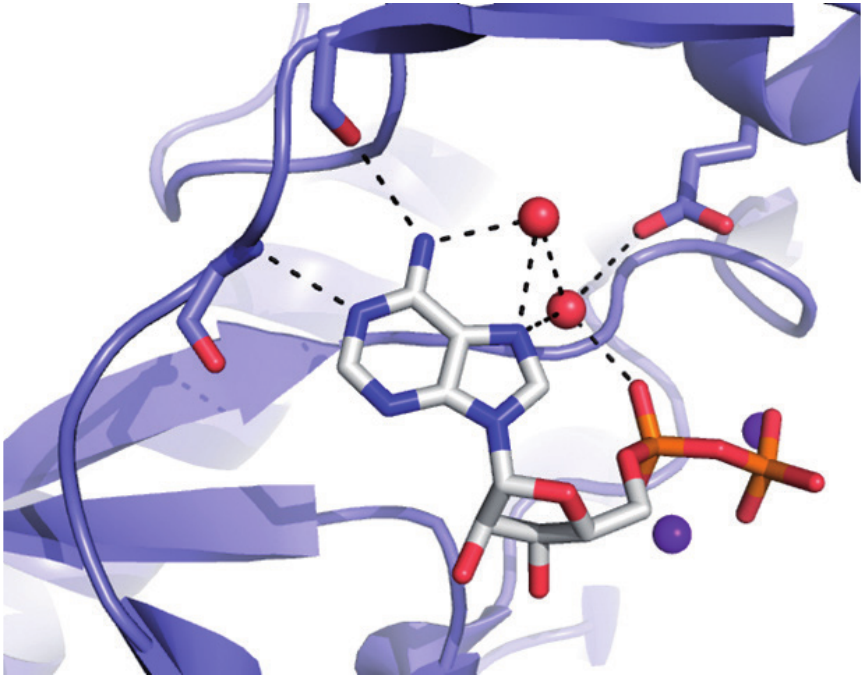
Bacterial antibiotic modifying enzyme: identification of the main reaction intermediate to be targeted.
Efficient and specific drugs directed against enzymes are much easier to design with a deep understanding of how the proteins work, and especially in which states they spend most of their time. Transient kinetics techniques (stopped-flow, quench-flow, cryoenzymology) are routinely used to measure protein-ligand kinetics (kon, koff ), identify reaction pathway and rate-limiting steps, as well as to characterize the effects of inhibitors thereon. Beside, other biophysical techniques are used to characterize drug-target interactions: classical steady state enzymology, isothermal titration calorimetry, thermal shift assay, molecular docking and X-ray crystallography. Optimized inhibitors are designed and their biological effects tested (in collaboration for cellular and animal models). The group focuses its research on new targets working on nucleos/tides for the design of innovative anti-infectious or anti-cancer therapies.
Collaborations: S. Pochet (I. Pasteur), P.A. Kaminsli (I. Pasteur), O. Dussurget (I. Pasteur), L. Chaloin (CNRS), R. Candau (UM), E. Serpersu (Univ. Tennessee), S. Kunzelmann (Crick Inst.).
References: Leban et al. Biochim Biophys Acta 2017; Kaplan et al. Biochim Biophys Acta 2016; Marton et al. J Med Chem 2015; Py et al. J Physiol 2015, Gráczer et al. FEBS J 2013; Labesse et al. Structure 2013.

